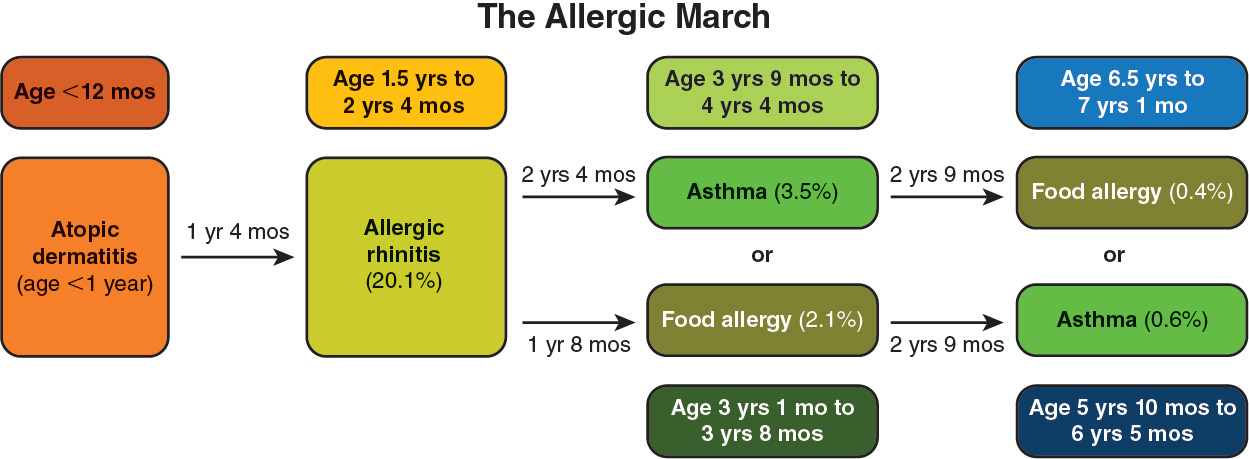
Treating atopic dermatitis may interrupt the allergic march.
Most patients with mild-to-moderate atopic dermatitis may be successfully treated with skin care, topical anti-inflammatory medications, and avoidance of skin irritants. See specific summary information on the treatment of atopic dermatitis.
Young children with severe or treatment-refractory atopic dermatitis may require other therapies if typical skin-care routines and topical anti-inflammatory medications are ineffective.
Emollients
Moisturizers are critical for the care of atopic dermatitis. The best moisturizers are ones that patients will use regularly. The American Academy of Allergy, Asthma and Immunology (AAAAI)/American College of Allergy, Asthma and Immunology (ACAAI) Joint Task Force on Practice Parameters (JTFPP) 2023 guidelines suggest not using prescription moisturizers. Rather, a fragrance-free over-the-counter moisturizer is preferable.
Emollients can also reduce the risk of epicutaneous sensitization. While the use of moisturizers may not prevent atopic dermatitis from ever manifesting, it can be used to prevent or reduce flares in patients with a genetic predisposition to atopic dermatitis. An American Academy of Pediatrics clinical report provides information on the use of emollients in preventing skin flares of infants and children who already have atopic dermatitis.
Topical Treatment
In patients with uncontrolled atopic dermatitis that is refractory to moisturizer alone, the following topical treatments are recommended:
- In cases of mild disease, calcineurin inhibitors can be used twice daily on small areas of skin for infants and children at least 3 months of age.
- Tacrolimus is approved for children at least 2 years of age, and for younger children it has been used off-label effectively, with a good safety profile.
- Pimecrolimus is a prescription, topical calcineurin inhibitor cream that can be used to treat mild-to-moderate atopic dermatitis.
- Topical corticosteroids can be used for atopic dermatitis in infants younger than 3 months. They are often recommended for use twice daily. Numerous topical corticosteroids of varying levels of potency are available.
- Topical phosphodiesterase 4 (PDE4) inhibitors, such as crisaborole 2% ointment, are recommended for mild-to-moderate dermatitis.
- In 2024, the US Food and Drug Administration (FDA) approved roflumilast cream 0.15% for mild-to-moderate atopic dermatitis in children at least 6 years of age. Roflumilast is a selective PDE4 inhibitor. Ongoing studies are looking at alternate dosing in younger age-groups.
- Topical Janus kinase (Jak) inhibitors, such as ruxolitinib, can be considered in certain patients at least 2 years of age. However, in the 2023 AAAAI/ACAAI JTFPP guidelines, in adolescent and adult patients with mild-to-moderate atopic dermatitis that was refractory to moisturization alone, the JTF panel suggested against adding topical ruxolitinib over continued usual care alone. Concerns about systemic absorption with topical Jak inhibitors are sufficient to limit the application of ruxolitinib to less than 20% body surface area. Additionally, the FDA has placed a boxed warning label on all Jak inhibitors owing to a study in rheumatoid arthritis with an oral pan-Jak inhibitor, tofacitinib.
- In December 2024, tapinarof cream was approved for children with atopic dermatitis aged at least 2 years. Tapinarof cream is an aryl hydrocarbon receptor agonist that suppresses the immune system primarily by interfering with inflammatory signaling via the NF-κB pathway.
- Wet wraps can be used with a topical low- to mid-potency steroid, for a limited period and body surface area.
Systemic Therapies
- Allergen immunotherapy is recommended in moderate and severe atopic dermatitis if the child’s condition is refractory or if the child has an intolerance or is unable to use mid-potency topical treatments.
- Biologics (monoclonal antibodies)
- With a high certainty of evidence, dupilumab is recommended in infants and children at least 6 months of age who have moderate or severe atopic dermatitis whose condition is refractory or who have an intolerance or are unable to tolerate mid- to high-potency topical treatments.
- With a high certainty of evidence, tralokinumab is recommended for use in children and adolescents at least 12 years of age who have moderate or severe atopic dermatitis and whose condition is refractory or who have an intolerance or are unable to tolerate mid- to high-potency topical treatments.
- Lebrikizumab was recently approved to treat moderate-to-severe atopic dermatitis in children and adolescents at least 12 years of age.
- Phototherapy treatment. Clinic-based, narrow-band UV-B treatment can be offered to older children with moderate-to-severe atopic dermatitis who are able to safely receive phototherapy treatment whose condition is refractory or who have an intolerance or are unable to tolerate mid- to high-potency topical treatments inclusive of an appropriate biologic agent.
- Oral Jak inhibitors are suggested, with low certainty of evidence, in patients with moderate-to-severe atopic dermatitis whose condition is refractory or who have an intolerance or are unable to tolerate mid- to high-potency topical treatments inclusive of an appropriate biologic agent.
- Agents include abrocitinib and upadacitinib. Agents vary in age of approval, between 12 and 18 years of age.
- As noted earlier, these agents carry an FDA boxed warning. The clinician must consider comorbidities, risk factors, values and preferences, and exceptional circumstances.
- Immunosuppressant therapy can be considered when other options are not optimal choices, are not tolerated, or are ineffective.
Recommendations against specific therapies for children include:
- Baricitinib 1 mg daily
- Azathioprine
- Cyclosporine
- Methotrexate
- Mycophenolate
- Systemic corticosteroids
Biologics and Risk Reduction for Allergic March
Biologics are molecularly targeted drugs derived from living organisms. Monoclonal antibodies are 1 type of biologic used to treat allergic disease.
Examples of biologics used to treat atopic disease in children in the United States are shown in the following table:
|
Biologic
|
Molecular Target |
Age |
Treatment Indication |
| Benralizumab |
IL-5Rα |
≥ 6 years |
Severe eosinophilic asthma |
| Dupilumab |
IL-4Rα (receptor for IL-4 and IL-13) |
≥ 6 months |
Atopic dermatitis (age range varies for its other childhood indications including asthma, EoE, chronic urticaria, chronic rhinosinusitis with nasal polyps) |
| Lebrikizumab-lbkz |
IL-13 |
≥ 12 years |
Moderate-to-severe atopic dermatitis |
| Mepolizumab |
IL-5 |
≥ 6 years |
Severe eosinophilic asthma; also approved to treat hypereosinophilic syndrome in ≥ 12 years of age |
| Nemolizumab-ilto |
IL-31Rα |
≥ 12 years |
Moderate-to-severe atopic dermatitis |
| Omalizumab |
IgE |
≥ 1 year |
Approved for IgE-mediated food allergy (≥ 1 year of age), asthma, and chronic idiopathic urticaria (≥ 12 years of age) |
| Tezepelumab |
|
≥ 12 years |
Severe asthma; targets thymic stromal lymphopoietin |
| Tralokinumab-ldrm |
IL-13 |
≥ 12 years |
Moderate-to-severe atopic dermatitis |
Dupilumab, which is approved for use in infants and children older than 6 months, is a humanized monoclonal antibody to the IL-4α receptor, which is activated by TH2 cytokines IL-4 and IL-13.
- TH2 differentiation is modulated by dupilumab.
- A detailed review of the effects of targeting the IL-4α receptor shows multiple mechanisms of immune regulation that contribute to its various clinical indications.
Dupilumab is the only biologic with evidence that it specifically interrupts the allergic march.
A 2024 observational cohort study demonstrated a reduced risk of allergic march progression over 3 years in children with atopic dermatitis who were prescribed dupilumab compared to conventional therapies.
- Children with atopic dermatitis who took dupilumab had a 32% reduced risk of allergic march progression compared to those using conventional therapies.
- Dupilumab had a 40% lower asthma risk and a 31% lower allergic rhinitis risk than conventional treatments over a 3-year observation period.
- The effect on food allergy was not significant.
A 2020 Cochrane review of biologic treatment for atopic dermatitis in pooled participants with an average of 32 years (range, 2 to 84 years) indicated that dupilumab was the most effective biological treatment for eczema.
- Dupilumab was more effective than placebo in achieving an eczema area severity index improvement of 75% and improvement in patient-oriented eczema measure score at 16 weeks (high‐certainty evidence).
- Longer-term effects compared with placebo had low-certainty evidence.
- Evidence for the effectiveness of most other immunosuppressive treatments for moderate-to-severe atopic eczema is of low certainty.
- Dupilumab use is associated with eye inflammation and eosinophilia.


![]()