General Questions
-
The AAP published its 2026 immunization schedule, which is based on the best available science in January 2026. A family-friendly version of the AAP schedule is also available on HealthyChildren.org.
-
Families may have many questions regarding the recent news regarding the CDC changes to the childhood immunization schedule. See this Clinician-Family Immunization Communication FAQ which provides clinicians with ways to frame conversations in response to these questions. Additional articles and resources on common immunization questions for families are available on www.healthychildren.org/immunizations in English and Spanish.
Coding and Payment
-
The shared clinical decision-making recommendation provides a payment pathway for vaccines that are no longer routinely recommended by the CDC. Additionally, the Vaccines for Children (VFC) program would continue to distribute vaccines that have a shared clinical decision making recommendation for those that want it.
-
Immunization coding is based on who provided counseling and when the counseling occurred:
- If counseling was provided by a physician, physician assistant, or nurse practitioner on the same date as the administration of the immunization, codes 90460 and 90461 representing counseling and administration would be reported. Multiple units of each code may be appropriate depending on how many different products are administered and the number of components in each injection.
- If counseling was provided by clinical staff or occurred on a previous date of service by a physician, physician assistant, or nurse practitioner such as when immunizations need to be split up on different dates for clinical reasons or if a practice has a separate immunization clinic day, codes 90471 and 90472 representing administration of the first injection and any additional injections administered would be reported.
When reporting codes 90460/90461 or 90471/90472, it’s important to remember that how you choose the codes and the number of units reported can differ based on the code set. If a physician, physician assistant, or nurse practitioner provides counseling on the same day as the immunization administration, the code selection is based on the number of components in the immunization given. However, if clinical staff provide counseling during a preventive visit or if the counseling is done by a physician, physician assistant, or nurse practitioner on a different day, then the code selection is based on the number of injections, or "per poke." See the AAP immunization coding table and preventive coding guide for additional information on reporting immunization administration.
-
Standalone immunization counseling utilizes three CPT codes: 90482, 90483, and 90484. The selection of the appropriate code is determined by the total time spent on counseling. It is important to note that only the time spent addressing refused immunizations can be included in the total time calculation. Additionally, some payers may require different codes instead of 90482, 90483, or 90484. For more information, refer to our factsheet on standalone immunization counseling.
When reporting counseling—whether it involves the administration of an immunization—it is essential to perform and document the following:
List all vaccine components- Who provided counseling, either a physician, a physician assistant, a nurse practitioner, or a clinical staff member
- Discussion of questions or concerns raised by parents or caregivers
- Reason for refusal of immunization(s)
A brief overview and providing the parent or caregiver with a vaccine information statement does not constitute immunization counseling according to CPT definitions.
Additional information about coding and payment for immunizations can be found on the AAP Vaccine Financing and Coding page. Please report any payer concerns or hassles to the Coding & Payment Hotline.
COVID
-
Each year, updated formulations of COVID vaccines are manufactured to provide better protection against currently circulating variants and prevent serious consequences of COVID-19, including severe disease, hospitalization, death, and long COVID.
The AAP recommends COVID-19 vaccination as follows:- Completion of an initial vaccination series for everyone ages 6 -23 months of age, or a single dose for those under age 2 years who previously completed their initial series.
- Two or more doses for children 6 months – 18 years of age who are moderately or severely immunocompromised.
- A single dose for all children and adolescents 2-18 years of age who are at high risk for severe COVID-19, residents of long-term care facilities or other congregate settings, persons who have never been vaccinated against COVID-19, or persons whose household contacts are at high risk for severe COVID-19
- Children 2-18 years of age whose parent or guardian desires protection from COVID-19 for their child should be offered a single dose.
There are two types of COVID-19 vaccines recommended for use in the United States – mRNA vaccines and the protein subunit vaccine. There is no preferential recommendation when multiple vaccine options are available.
Please see the AAP Pediatric COVID-19 Vaccine Dosing Quick Reference Guide for more details, including recommendations on the intervals between doses.
Additional Resources:
-
AAP recommends a single dose of COVID-19 vaccine for all children and adolescents 2-18 years of age who are at high risk for severe COVID-19 (other than moderately or severely immune compromised children, who are recommended to receive multiple doses). These risk groups are described in the AAP Policy Statement as follows:
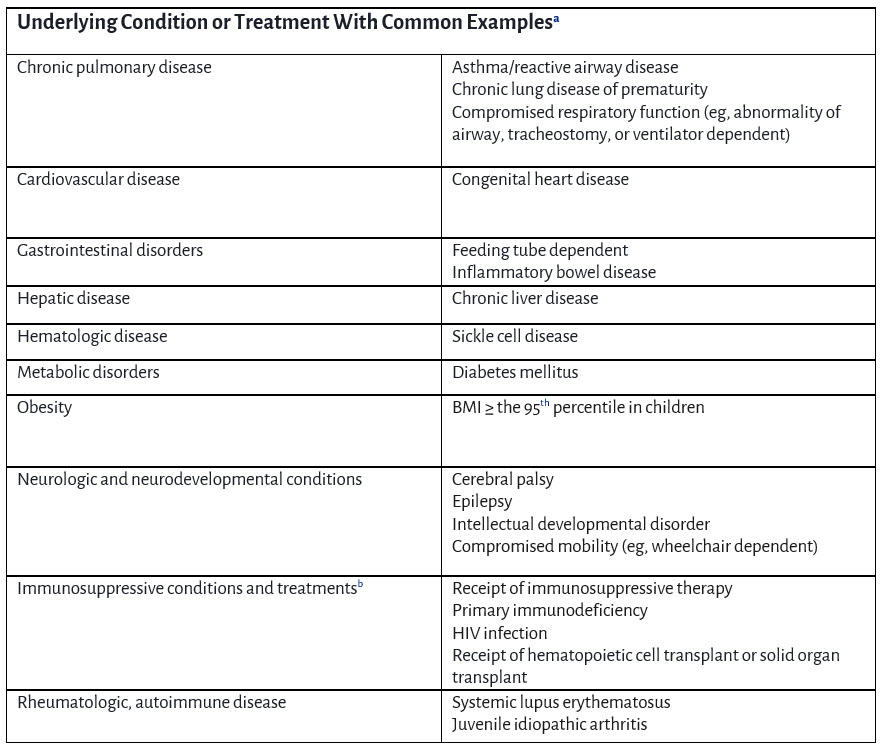 a List of examples is not exhaustive
a List of examples is not exhaustive
b Children who are moderately or severely immunocompromised require 2 or more doses of COVID-19 vaccine. Refer to AAP Recommended Child and Adolescent Immunization Schedule or AAP Pediatric COVID-19 Vaccine Dosing Quick Reference Guide for dosing guidance. -
Moderna mRNA Vaccine
- Spikevax Package Insert (ages 6 months and up)
- mNEXSPIKE Package Insert (ages 12 years and up)
Pfizer-BioNTech mRNA Vaccine
- Comirnaty Package Insert (ages 5 years and up)
Novavax Protein Subunit Vaccine
- Nuvaxovid Package Insert (ages 12 years and up)
-
Please refer to the AAP Pediatric COVID-19 Vaccine Dosing Quick Reference Guide for dosing intervals. This resource offers the dosing intervals for unvaccinated children and previously vaccinated children. Also included is dosing information for children who are moderately or severely immunocompromised.
-
A child should receive the recommended dosing based on their age at the START of their vaccine series. For example, a child who begins their initial series at age 23 months should receive two doses of Moderna Spikevax vaccine.
-
A child should receive age-appropriate vaccine product and dosage based on their age on the day of vaccination. For example, for children who turn 11 to 12 years of age, administer the recommended product/dosage for 12 year olds for all doses that are administered after the child turns 12 years of age See additional information in the AAP Pediatric COVID-19 Vaccine Dosing Quick Reference Guide.
-
COVID-19 vaccination should be deferred for patients with a current SARS-CoV-2 infection until they recover from their acute illness.
Individuals who recently had SARS-CoV-2 infection may consider delaying COVID-19 vaccine by 3 months from symptom onset or positive test (if infection was asymptomatic). Studies have shown that increased time between infection and vaccination may result in an improved immune response to vaccination. -
No, the side effect and safety profile are the same as that of previous formulations. The side effects have been similar to other routine vaccines- sore arm or leg, redness, fatigue, fever, chills, headache, myalgia, and arthralgia. The side effects are temporary and mostly mild or moderate. Anaphylaxis has been observed following receipt of COVID-19 mRNA vaccines, but this has been rare. Rare cases of myocarditis or pericarditis have been reported – most often in males between 12-39 years of age. Additional information on common side effects and rare adverse reactions can be found in the package inserts of the vaccine products:
Moderna mRNA Vaccine
- Spikevax Package Insert (ages 6 months and up)
- mNEXSPIKE Package Insert (ages 12 + years)
Pfizer-BioNTech mRNA Vaccine
- Comirnaty Package Insert (ages 5+ years)
Novavax Protein Subunit Vaccine
- Nuvaxovid Package Insert (ages 12+ years)
-
Healthcare providers are required by law to report to Vaccine Adverse Event Reporting System (VAERS):
- Any adverse event listed in the VAERS Table of Reportable Events Following Vaccination that occurs within the specified time period after vaccination
- An adverse event listed by the vaccine manufacturer as a contraindication to further doses of the vaccine
Healthcare providers are strongly encouraged to report:
- Any adverse event that occurs after the administration of a vaccine licensed in the United States, whether or not it is clear that a vaccine caused the adverse event
- Vaccine administration errors
Hepatitis B
-
The AAP’s recommendations regarding hepatitis B vaccination have not changed in response to the CDC immunization schedule changes. The AAP continues its current recommendation for the birth dose of hepatitis B vaccine for newborns within the first 24 hours of life. Additionally, AAP continues to recommend subsequent hepatitis vaccine doses between 1-2 months and 6-18 months.
- AAP Immunization Schedule
- Elimination of Perinatal Hepatitis B: Providing the First Vaccine Dose Within 24 Hours of Birth | Pediatrics | American Academy of Pediatrics
- AAP Red Book Chapter: Hepatitis B
-
The AAP does not recommend serology testing for infants to guide further Hep B vaccination, as the significance of Hep B sAb level of >10 mIU/mL in early infancy is unknown. There is also insufficient evidence regarding the significance of antibody levels obtained mid-series and the relationship with long-term immunity against hepatitis B, particularly in this age group. These laboratory results would not reliably represent immune status or a scientific basis for a decision to continue or forego subsequent vaccination. This laboratory test represents an unnecessary blood test and procedure for infants, a barrier for families, additional costs, a risk for immunity delay, and other potential harms to infants without conferring value for evidence-based decision making in a clinical setting.
-
- Hepatitis B infection in infancy results in more serious disease burden: Around 90% of newborns infected with hepatitis b virus (HBV) develop chronic infection, compared to < 5% of adults infected later in life. But the birth dose eliminates this risk. People who were infected with hepatitis B at birth and are not treated have up to a 25% lifetime risk of developing liver cancer.
- Chronic hepatitis B infection remains a major health issue, as roughly 660,000 people in the US live with chronic hepatitis b infection and are often undiagnosed. This infection can easily be transmitted to others as the virus can live on surfaces outside of the body for more than 7 days.
- Risk-based screening has proven unsuccessful in the US, as there are real-world gaps and limitations in screening. Current data show that 12-18% of pregnant people don’t receive hepatitis B surface antigen (HBsAg) testing and only 35% of those who test positive receive all recommended follow-up care. Additionally, about 35-65% of HBsAg-positive mothers had no identifiable risk factors that would flag them for targeted screening.
- Universal screening works and the vaccine is safe. The universal hepatitis B vaccination program has resulted in a 99% decline in pediatric cases. The hepatitis B vaccinees have undergone randomized controlled trials including those with placebo-controlled designs, which have been summarized by CIDRAP’s Vaccine Integrity Project. Additionally, the vaccine has a long-standing and proven safety record.
-
Risk-based strategies were tried throughout the 1980s in the US and failed to reduce incidence of infant infection. CDC data showed that 30–40% of hepatitis B patients had no identifiable risk factors, meaning targeted approaches would never reach them. Similarly, 35–65% of HBsAg-positive mothers had no identifiable risk factors and would never be flagged under targeted screening programs.
Implementation of an effective risk-based vaccination strategy based on maternal screening results is dependent upon universal maternal screening of HBsAg. Current data show that 12–18% of pregnant women don't receive HBsAg testing. Of those who test positive, only 35% receive all recommended follow-up care. The fragmented US healthcare system is not set up to catch everyone through risk-based vaccination approaches. The US shifted to universal infant vaccination in 1991, which has resulted in a 99% decrease in infant hepatitis infections.
Countries without a universal birth dose recommendation have smaller populations, high rates of universal prenatal screening, and high rates of adherence to recommended follow-up care.
-
The following are key steps appropriately administering the birth dose of hepatitis B vaccine based on AAP policy:
- Identify HBsAg-positive mothers before delivery and document maternal HBsAg status in infant records;
- Resolve unknown HBsAg status of mothers as soon as possible around delivery, and document maternal status in infant records;
- For all infants born to HBsAg-positive mothers, administer both hepatitis B vaccine and hepatitis B immune globulin (HBIG) within 12 hours of birth, regardless of any maternal antenatal treatment with antiviral medications;
- For all infants with birth weight greater than or equal to 2000 g born to HBsAg-negative mothers, administer hepatitis B vaccine as a universal routine prophylaxis within 24 hours of birth;
- For all infants with birth weight less than 2000 g born to HBsAg-negative mothers, administer hepatitis B vaccine as a universal routine prophylaxis at 1 month of age or at hospital discharge (whichever is first);
- For all infants born to HBsAg-unknown mothers, administer hepatitis B vaccine within 12 hours of birth, and:
- For infants with birth weight greater than or equal to 2000 g, administer HBIG by 7 days of age or by hospital discharge (whichever occurs first) if maternal HBsAg status is confirmed positive or remains unknown;
- For infants with birth weight less than 2000 g, administer HBIG by 12 hours of birth unless maternal HBsAg status is confirmed negative by that time;
- Document infant vaccination accurately in birth hospital records and in the appropriate CDC Immunization Information Systems and state immunization registry. Review documentation accuracy periodically and address identified errors.
-
- Why Do Babies Need the Hepatitis B Vaccine? - HealthyChildren.org is an article for families and is available in Spanish
- Fact Checked: Hepatitis B Vaccine Given to Newborns Reduces Risk of Chronic Infection summarizes key facts about the importance of the birth dose
- Maternal and Infant Immunization Discussion Guides include talking points for clinicians and family-friendly infographics about Hepatitis B and other recommended immunizations that can be shared during patient encounters or outside the clinical visit
Measles
-
The AAP recommends routine vaccination against measles and early vaccination during special circumstances.
The AAP recommends individuals receive 2 doses of measles, mumps, rubella (MMR) or measles, mumps, rubella and varicella (MMRV) vaccine. Routine timing for these vaccines are at the following ages
- Dose 1: 12 – 15 months
- Dose 2: 4 – 6 years
The minimum interval is 28 days for MMR and 90 days for MMRV.
-
Vaccination is the most effective way to prevent measles. A dose of measles vaccine administered after 12 months of age results in immunity in 93% of people. The 2nd dose increases immunity to 97%.
-
Since the introduction of measles vaccine in the United States in 1963, with millions of MMR doses being administered, transmission of measles through the MMR vaccine has not occurred for anyone (immunocompetent or immunocompromised).
-
The routine vaccination series is for the 1st dose of MMR to be administered at 12-15 months of age with the 2nd dose at 4-6 years of age. See information below on domestic measles outbreaks from the AAP Red Book.
Health care providers should follow vaccination recommendations issued by the state, local, tribal, or territorial health departments for areas experiencing sustained, community-wide measles transmission and an ongoing risk of exposure. In some cases, additional vaccinations may be recommended beyond the routine MMR vaccination schedule. For example, health departments may recommend:
- A second dose of MMR vaccine for preschool-aged children aged 1 to 4 years who received one prior dose and live in or plan to travel to the outbreak area. Children with no documentation of vaccination history should receive two doses, at least 28 days apart.
- An early dose of MMR vaccine for infants aged 6–11 months who live in or are traveling to the outbreak area.
- Providers should weigh the benefit of protection from measles during an outbreak against the risk of decreased immune responses in infants vaccinated with MMR before 12 months of age.
- Infants younger than 12 months of age are at greatest risk of severe illness. Vaccination of infants aged 6–11 months minimizes the risk of disease and death that could occur in these infants during measles outbreaks.
- The level of protective antibodies is lower and may remain lower in children vaccinated at younger than 12 months of age than in children vaccinated later. Infants who receive one dose of MMR vaccine before their first birthday should receive two more doses according to the routinely recommended schedule (one dose at 12 through 15 months of age and another dose at 4 through 6 years of age or at least 28 days later). (For additional information, see: 2025_providerletter_mmrtravelandoutbreakrecs.pdf section: “Immune Response to Measles Vaccination”.)
Vaccination of visitors to outbreak-affected areas should be consistent with the guidance of the state, local, tribal, or territorial health department for residents of the outbreak-affected community. For example, if no vaccination recommendation was made by the local health department for infants aged 6–11 months living in the outbreak community, then vaccination of infant travelers visiting the outbreak area would also not be recommended. Healthcare providers can find updated outbreak recommendations issued by state or local health departments on local web sites or the Red Book Online Outbreaks page.
MMR can be used as post-exposure prophylaxis if administered < 72 hours after measles exposure in individuals 6 months of age and older who do not have evidence of measles immunity.
-
Providers should weigh the benefit of protection from measles during an outbreak against the risk of decreased immune responses in infants vaccinated with MMR before 12 months of age.
Infants younger than 12 months of age are at greatest risk of severe illness. Vaccination of infants aged 6–11 months minimizes the risk of disease and death that could occur in these infants during measles outbreaks.
The level of protective antibodies is lower and may remain lower in children vaccinated at younger than 12 months of age than in children vaccinated later. Infants who receive one dose of MMR vaccine before their first birthday should receive two more doses according to the routinely recommended schedule (one dose at 12 through 15 months of age and another dose at 4 through 6 years of age or at least 28 days later). (View additional information, see section “Immune Response to Measles Vaccination”.)
-
AAP is not aware of claims denials for early MMR vaccination following public health guidelines. Please contact the AAP Coding and Payment Hotline for support in addressing coding and payment-related questions or if you experience a payment denial.
-
According to Immunize.org, adults are considered to have presumptive evidence of immunity to measles if they have written documentation of at least 1 dose of MMR vaccine on or after their 1st birthday, written documentation of 2 doses of MMR vaccine for individuals at high risk, laboratory evidence of measles immunity, laboratory confirmation of measles infection, or birth before 1957.
During measles outbreaks, health departments may provide additional recommendations to protect their communities, including a second dose of MMR for adults who have received only 1 dose previously. During an outbreak of measles in a healthcare facility, or in healthcare facilities serving a measles outbreak area, two doses of MMR vaccine are recommended for healthcare personnel, regardless of birth year, who lack other presumptive evidence of measles immunity. There are no recommendations to receive a third dose of MMR vaccine during measles outbreaks.
-
Please see Measles FAQ’s for additional information specific to measles infection prevention and control.
RSV Immunization
-
The AAP recommends RSV immunization with nirsevimab or clesrovimab shortly before or during RSV season for:
- infants <8 months of age born during or entering their first RSV season if:
- pregnant parent did not receive RSVpreF vaccine during this pregnancy,
- pregnant parent’s RSVpreF vaccination status is unknown, or
- infant was born <14 days after the pregnant parent’s RSVpreF vaccination
The AAP recommends RSV immunization with nirsevimab only shortly before or during RSV season for:
- infants and children 8 through 19 months of age at high risk of severe RSV disease and entering their second RSV season, regardless of the RSV vaccination status of the pregnant parent or the child’s prior receipt of nirsevimab or clesrovimab when <8 months of age. High-risk criteria include the following:
- Children with chronic lung disease of prematurity who required medical support (chronic corticosteroid therapy, diuretic therapy, or supplemental oxygen) at any time during the 6-month period before the start of the second RSV season
- Children with severe immunocompromise
- Children with cystic fibrosis who have either:
- manifestations of severe lung disease (previous hospitalization for pulmonary exacerbation in the first year of life or abnormalities on chest imaging that persist when stable), or
- weight-for-length that is less than the 10th percentile
- American Indian or Alaska Native children
Note that all ages refer to chronologic age, not corrected age.
- infants <8 months of age born during or entering their first RSV season if:
-
The AAP does not recommend RSV immunization for most infants born to a mother who received maternal RSV vaccine during their most recent pregnancy. The exception is infants where less than 14 days have elapsed between vaccination and birth. In this situation, the infant should receive nirsevimab or clesrovimab per AAP recommendations.
RSV immunization can be considered for infants born to a mother who received maternal RSV vaccine when, per the clinical judgement of the healthcare provider, the potential incremental benefit of administration is warranted, including but not limited to the following rare circumstances:
- Infants born to pregnant people who may not mount an adequate immune response to vaccination or have conditions associated with reduced transplacental antibody transfer
- Infants who have undergone cardiopulmonary bypass or extracorporeal membrane oxygenation leading to loss of maternal antibodies
- Infants with substantial increased risk for severe RSV disease (eg, hemodynamically significant congenital heart disease, intensive care admission and requiring oxygen at discharge)
-
RSV monoclonal antibody should be administered during October through the end of March in most of the continental United States. However, recommendations on the timing of RSV antibody administration are intentionally flexible to optimize patient access, including insurance payment. For example, in Alaska RSV circulation patterns are less predictable, and the duration of the RSV season is often longer than the national average. Tropical climates might also have RSV circulation patterns that differ from most of the continental US or that are unpredictable. In addition, because the timing of RSV activity varies geographically in other regions of the US, public health authorities may elect to provide revised guidance regarding the timing of RSV antibody administration based on local surveillance data and feasibility of implementation (ie, extending or shortening the recommended administration period of October–March). Public health authorities should consider the advantages and disadvantages of modifying the timing of administration. Providers, including regional medical centers and health systems, should consult with state or territorial health departments before systematically modifying the recommended months for RSV antibody administration for their eligible patient populations. For more information, including an outline of potential advantages and disadvantages of modifying timing of administration, see http://dx.doi.org/10.15585/mmwr.mm7432a3.
Healthcare providers may choose to give nirsevimab or clesrovimab before the start of RSV season if they feel that the child may not return for a visit when RSV immunization would be recommended. For example, a clinician may choose to give RSV immunization to an infant who presented for care in September who has not yet received a dose of RSV immunization and may be unlikely to return for a visit in October or November. Nirsevimab and clesrovimab have been shown to protect against severe RSV disease for at least 5 months, and the ideal timing of administration may differ depending on the clinical situation.
-
Nirsevimab and clesrovimab are monoclonal antibody products that are passive immunizations. While not technically “vaccines” in a traditional sense (active immunization), they are being used in a manner similar to routine childhood vaccines and may be referred to as vaccines by some entities.
Nirsevimab and clesrovimab confer long-lasting protection from RSV, with protection expected to last at least 5 months (about the length of a typical RSV season). Nirsevimab and clesrovimab are part of the Vaccines for Children program.
-
The highest risk for severe RSV is in children under 6 months of age. Infants 8 months and older will be entering their second RSV season and have likely already experienced their first RSV infection and will not receive the full benefits of RSV immunization.
-
Yes. Optimal timing for administration is within 1 week after birth during the RSV season. Administering RSV immunization through the end of the season is important because the risk of severe disease is highest during the first few months of life and even when the season is over, RSV infections still occur, but at a lower incident rate. Additionally, being a monoclonal antibody product, RSV immunization provides immediate protection to the infant.
-
Prior infection should generally not be used to determine an infant’s eligibility for RSV immunization.
-
Protection from RSV immunization is expected to last at least 5 months, about the length of an RSV season, and is expected to reduce the risk of severe RSV disease by about 80%. Data from the 2024-2025 RSV season showed that nirsevimab was effective against RSV-associated emergency department encounters and hospitalization among infants in their first RSV season. Specifically, data from the VISION Multi-Site Network of Electronic Health Records of 160 emergency departments and 131 hospitals showed nirsevimab was 63% effective at preventing RSV-associated ED encounters and 79% effective at preventing RSV-associated hospitalization.
-
In accordance with Red Book guidance, simultaneous administration of RSV immunization with age-appropriate vaccines is recommended. CDC does not address the issue of maximum volumes that can be injected into each muscle group in different age groups. Based on discussions with CDC, AAP offers the suggested volumes as follows:
- Deltoid: Average 0.5 mL (range 0.5–2 mL)
- Vastus Lateralis: Average 1–4 mL (range 1–5 mL)
Infants and toddlers would fall at the lower end of the range, whereas adolescents and adults would generally fall on the higher end of the range. Strategies healthcare providers can use to decrease the number/injection volume include:
- Healthcare providers should always use professional judgement when administering injections. Muscle size can vary greatly from one patient to another.
- Include an age-appropriate combination vaccine in the facility’s inventory (Pentacel, Pediarix, Vaxelis).
- Use an alternate route (other than IM) if possible. IPV (single component, NOT a combination vaccine), MMR (toddlers and infant travelers only), Varicella-containing vaccines (toddlers only), and PPSV23 (high-risk toddlers only) can be administered subcutaneously.
- Take advantage of recommended age ranges some of the routinely recommended vaccines have. For example, the 3rd dose of HepB can be given as late as 18 months of age to children born to mothers who are HBsAg negative.
-
Both nirsevimab and clesrovimab are contraindicated in infants and young children with a history of serious hypersensitivity reactions, including anaphylaxis, to nirsevimab or clesrovimab or to any of their components. Illness or febrile diseases are not contraindications to receiving an RSV immunization. The AAP suggests following Immunize.org guidance, which recommends that vaccination should be deferred for persons with a moderate or severe acute illness, as this precaution avoids causing diagnostic confusion between the underlying illness and potential adverse effects of immunization. Similar to routine childhood vaccines, mild illness – with or without fever – should not be used as a reason to delay administration of nirsevimab or clesrovimab.
-
Immunize.org published an Immunization Information Statement (VIS-like document) for families. It states that “After getting an RSV preventive antibody, your child might have temporary pain, redness, swelling where the injection was given, or a rash.” In addition, the nirsevimab package insert and clesrovimab package insert have additional information on adverse reactions. For nirsevimab, “Most common adverse reactions were rash (0.9%) and injection site reactions (0.3%).” For clesrovimab, “Most common adverse reactions were injection-site erythema (3.8%), injection site swelling (2.7%), and rash (2.3%).
-
Adverse events when giving an RSV immunization alone should be reported to the FDA’s MedWatch Adverse Event Reporting Program.
If an adverse event occurs while co-administering RSV immunization with a vaccine, it should be reported to the Vaccine Adverse Event Reporting System.
-
Please see RSV FAQ’s for additional Q&A’s on administration of RSV immunization.
Last Updated
02/24/2026
Source
American Academy of Pediatrics